
As a rheumatologist in the Division of Rheumatology and physician-scientist in the Ludwig Center for Cancer Genetics and Therapeutics at Johns Hopkins, Dr. Konig’s research focuses on chimeric T cell- and antibody-based strategies for the treatment of rheumatic diseases and cancer. A primary goal of his translational research program is the development of antigen-specific and personalized immunotherapies for autoimmune rheumatic diseases, with the intent to achieve sustained disease remission and a functional cure.
Dr. Konig has a long-standing interest in mechanisms of disease initiation and propagation of autoimmune rheumatic and inflammatory diseases. His postdoctoral research elucidated how specific bacteria may cause loss of immunological tolerance and the development of anti-citrullinated protein antibodies (ACPAs) in rheumatoid arthritis, a hallmark of this autoimmune disease. His work identified Aggregatibacter actinomycetemcomitans as a bacterial pathogen that induces hypercitrullination and the release of citrullinated autoantigens from neutrophils during periodontal infection. These mechanistic findings may explain the long-known clinical association of rheumatoid arthritis and gum disease and prove critical to research efforts to prevent the development of rheumatoid arthritis in high-risk individuals who have evidence of preclinical autoimmunity.

In Simple Terms: Information for Patients
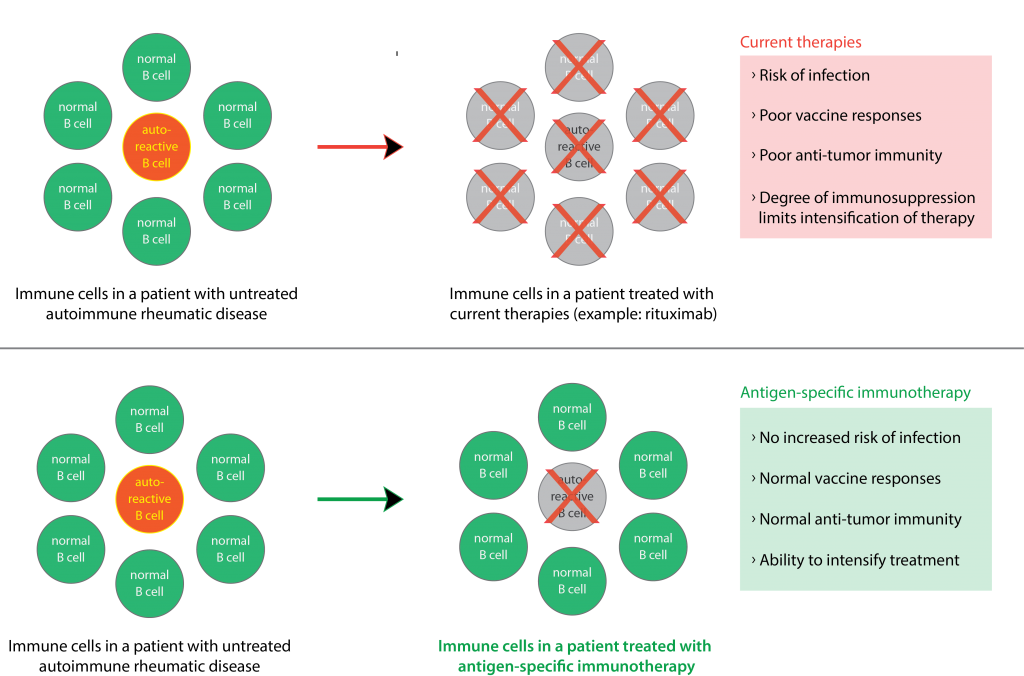
Curative treatment approaches for autoimmune diseases do currently not exist. All current strategies in treating rheumatic disease rely on broadly acting immunosuppressive drugs that suppress self-reactive (harmful) but also all protective immune responses, resulting in risk of infection, inadequate vaccine responses, and treatment-related morbidity.
Ideal treatments for autoimmune diseases would eliminate only the self-targeting immune cells that cause rheumatic disease symptoms and damage but not affect protective and beneficial immune responses. Our goal is to develop such personalized and antigen-specific immunotherapies.
Chimeric antigen receptor (CAR) and related T cell therapies have already revolutionized the treatment of blood cancers over the past decade and can achieve sustained cancer-free remission. Our lab uses similar tools (i.e., CRISPR technology and cell engineering) to reprogram a patient’s T cells to allow them to specifically eliminate autoreactive immune cells – while preserving all normal immune cell populations. If successful, such therapies have the potential to fundamentally change the treatment of patients with rheumatic diseases over the next decade and provide us with new cell-based therapies and drugs that can achieve sustained disease remission or even functional cure without increasing the risk of opportunistic infection. We believe that we already today have the tools to achieve this goal.
I would like to donate to accelerate the development of new therapies for rheumatic diseases
If you would like to donate to this cause specifically, please select “Other” under the “Please designate my gift to support” drop-down menu and specify “Konig Lab”.
Visit our website
I am interested in more information/ joining the lab

Selected Publications
- Hwang MS, Mog BJ, Douglass J, Pearlman AH, Hsiue EH, Paul S, DiNapoli SR, Konig MF, Pardoll DM, Gabelli SB, Bettegowda C, Papadopoulos N, Vogelstein B, Zhou S, Kinzler KW. Targeting loss of heterozygosity for cancer-specific immunotherapy. Proc Natl Acad Sci U S A. 2021 Mar 23;118(12). PubMed PMID: 33731480; PubMed Central PMCID: PMC8000272.
- Paul S, Pearlman AH, Douglass J, Mog BJ, Hsiue EH, Hwang MS, DiNapoli SR, Konig MF, Brown PA, Wright KM, Sur S, Gabelli SB, Li Y, Ghiaur G, Pardoll DM, Papadopoulos N, Bettegowda C, Kinzler KW, Zhou S, Vogelstein B. TCR β chain-directed bispecific antibodies for the treatment of T cell cancers. Sci Transl Med. 2021 Mar 10;13(584):eabd3595. PMID: 33649188
- Hsiue EH, Wright KM, Douglass J, Hwang MS, Mog BJ, Pearlman AH, Paul S, DiNapoli SR, Konig MF, Wang Q, Schaefer A, Miller MS, Skora AD, Azurmendi PA, Murphy MB, Liu Q, Watson E, Li Y, Pardoll DM, Bettegowda C, Papadopoulos N, Kinzler KW, Vogelstein B, Gabelli SB, Zhou S. Targeting a neoantigen derived from a common TP53 mutation. Science. 2021 Mar 5;371(6533):eabc8697. PMID: 33649166; PMCID: PMC8208645
- Douglass J, Hsiue EH, Mog BJ, Hwang MS, DiNapoli SR, Pearlman AH, Miller MS, Wright KM, Azurmendi PA, Wang Q, Paul S, Schaefer A, Skora AD, Molin MD, Konig MF, Liu Q, Watson E, Li Y, Murphy MB, Pardoll DM, Bettegowda C, Papadopoulos N, Gabelli SB, Kinzler KW, Vogelstein B, Zhou S. Bispecific antibodies targeting mutant RAS neoantigens. Sci Immunol. 2021 Mar 1;6(57). PubMed PMID: 33649101; PubMed Central PMCID: PMC8141259.
- Giles JT, Reinholdt J, Andrade F, Konig MF. Associations of Antibodies Targeting Periodontal Pathogens With Subclinical Coronary, Carotid, and Peripheral Arterial Atherosclerosis in Rheumatoid Arthritis. Arthritis Rheumatol. 2021 Apr;73(4):568-575. PMID: 33205531; PMCID: PMC8005413
- Konig MF, Powell M, Staedtke V, Bai RY, Thomas DL, Fischer N, Huq S, Khalafallah AM, Koenecke A, Xiong R, Mensh B, Papadopoulos N, Kinzler KW, Vogelstein B, Vogelstein JT, Athey S, Zhou S, Bettegowda C. Preventing cytokine storm syndrome in COVID-19 using α-1 adrenergic receptor antagonists. J Clin Invest. 2020 Jul 1;130(7):3345-3347; PMCID: PMC7324164
- Konig MF. The microbiome in autoimmune rheumatic disease. Best Pract Res Clin Rheumatol. 2020 Feb;34(1):101473. Review. PubMed PMID: 32044247; PubMed Central PMCID: PMC7295668.
- Konig MF, Abusleme L, Reinholdt J, Palmer RJ, Teles RP, Sampson K, Rosen A, Nigrovic PA, Sokolove J, Giles JT, Moutsopoulos NM, Andrade F. Aggregatibacter actinomycetemcomitans-induced hypercitrullination links periodontal infection to autoimmunity in rheumatoid arthritis. Sci Transl Med. 2016 Dec 14;8(369):369ra176. PMID: 27974664; PMCID: PMC5384717
- Konig MF, Giles JT, Nigrovic PA, Andrade F. Antibodies to native and citrullinated RA33 (hnRNP A2/B1) challenge citrullination as the inciting principle underlying loss of tolerance in rheumatoid arthritis. Ann Rheum Dis. 2016 Nov;75(11):2022-2028. PubMed PMID: 26865600.
- Konig MF, Paracha AS, Moni M, Bingham CO 3rd, Andrade F. Defining the role of Porphyromonas gingivalis peptidylarginine deiminase (PPAD) in rheumatoid arthritis through the study of PPAD biology. Ann Rheum Dis. 2015 Nov;74(11):2054-61. PubMed PMID: 24864075; PubMed Central PMCID: PMC4368502.
View the complete list of publications by Dr. Konig here.


